Apricus Biosciences, Inc. (NASDAQ: APRI)
Apricus Biosciences, Inc. (APRI), a biopharmaceutical company today announced it has entered a license agreement with Allergen for the U.S rights to Vitaros. Vitaros is a topical cream used for the treatment of erectile dysfunction (ED), and is a new entrant into the U.S. ED market.
Apricus Biosciences, Inc. CEO’s Comments
Richard Pascoe, Chief Executive Officer of Apricus, commented,
“We are pleased to announce this transaction for the return of the U.S. Vitaros rights to Apricus and we look forward to working with Allergan to transition the regulatory submission back within our organization. This agreement creates a framework where both parties can leverage their respective strengths with the goal of bringing this novel topical treatment to the world’s largest ED market, which had sales of over $3 billion in 2014 according to IMS Health. Moreover, we believe that with Apricus’ broad Vitaros expertise and internal know-how, coupled with our proven success in obtaining regulatory approvals for Vitaros in other territories, we are well equipped to pursue regulatory approval for Vitaros in the U.S.” Globe Newswire
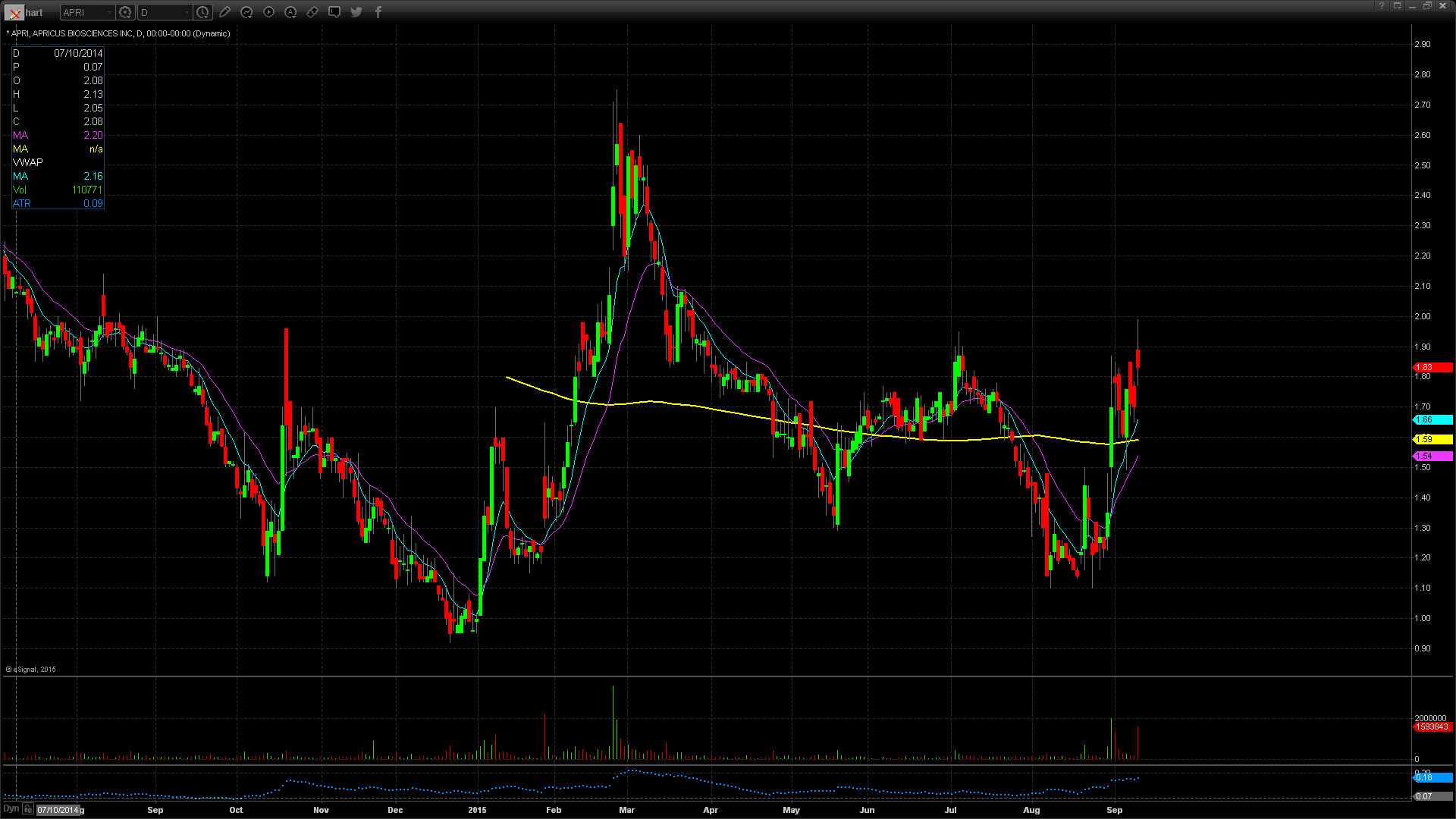
APRI Technical Analysis
APRI gapped up in price to $1.89 yesterday from a prior close of $1.70 which is a 10% increase in price based on favorable news. Taking a look at the daily chart we can see the last time the stock traded at this price level was back on March 26 when it traded at $1.93. Taking a further look back we can see the stock reached its 52 week high of $2.75 only a month prior to that on Feb 25. In the pre market the stock did reach a high of $2.25 which means that it gave back $0.36 at the open which is equivalent to 16%. This concerns me a little bit as the stock may be overextended a bit at the current price level. For trading purposes I would the whole number of $2.00 as my entry point looking for a pop through it and a run to the pre market highs of $2.25. My stop loss price would be the gap up price of $1.89 fearing anything below that and the stock will start to fill in the gap up.
Company Profile
Apricus Biosciences, Inc. engages in developing and commercializing products and product candidates in the areas of urology and rheumatology in the United States, Canada, Europe, and internationally. The companys lead product is Vitaros, a topically-applied cream formulation of alprostadil used for the treatment of erectile dysfunction. Its marketing partners for Vitaros include Abbott Laboratories Limited, Takeda Pharmaceuticals International GmbH, Hexal AG (Sandoz), Recordati Ireland Ltd. (Recordati S.p.A.), Bracco S.p.A. and Laboratoires Majorelle. The companys products under development include second-generation Vitaros, a proprietary stabilized dosage formulation that is expected to be stored at room temperature conditions; and RayVa, which is in Phase IIa clinical trial for the treatment of Raynauds Phenomenon. It also plans to initiate a Phase IIb trial for fispemifene, a tissue-specific selective estrogen receptor modulator to treat secondary hypogonadism, chronic prostatitis, and lower urinary tract symptoms in men; and out-license Femprox, a product candidate for the treatment of female sexual interest/arousal disorder. The company was formerly known as NexMed, Inc. and changed its name to Apricus Biosciences, Inc. in September 2010. Apricus Biosciences, Inc. was founded in 1987 and is headquartered in San Diego, California. Yahoo Finance