Genocea Biosciences, Inc. (NASDAQ: GNCA)
Genocea Biosciences, Inc. (GNCA), a biopharmaceutical company yesterday announced positive top line results from its phase 2b clinical trial for GEN-003 in patients with genital herpes.
The study showed statistically significant expected Phase 3 primary endpoint results with Phase 3 dose.
Positive results were reached on multiple secondary endpoints. These results are important as this could possibly be the first new treatment for genital herpes in the past 20 years a disease affecting millions of people.
Genocea Biosciences, Inc. CEO’s Comments
Chip Clark, President and CEO of Genocea, commented: “We believe these data further solidify the strong clinical profile for GEN-003, which could provide durable, convenient efficacy to a large and, we believe, highly dissatisfied patient population and serve as a cornerstone treatment of this burdensome disease.” Globe Newswire
GNCA Technical Analysis
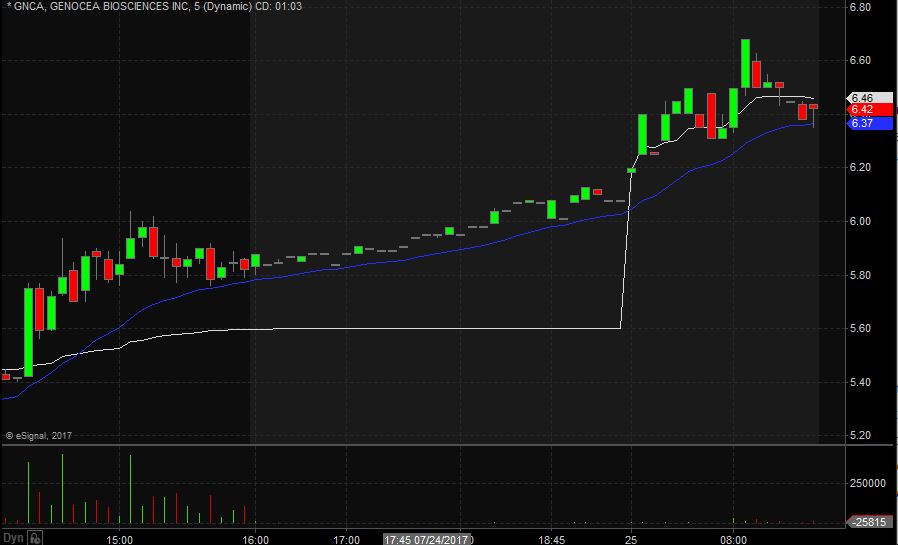
GNCA opened trading yesterday at $5.59 which was up from the previous day’s trading close of $5.05. GNCA closed trading yesterday at $5.83 and spiked up after market to $6.08, equivalent to a 20% increase from the prior day’s closing price. Taking a look at the daily chart we can see the last time GNCA above below these levels we have to go back to June 8th when it traded at lows of $6.13.
Taking a closer look at the daily chart we can see that before the spike up GNCA had been in an overall downward trend dating back to April 24th when it traded at $7.10. GNCA has a float of 22.76 million shares and traded 31.86 times the normal daily trading volume on Monday.
For day trading purposes, I would like to see GNCA open trading on Tuesday above $5.75 and if it does I would be looking to take a long position at the bell. My stop loss would be $0.15 from my entry position fearing anything more than that and the stock would start to fill in the gap up.
Company Profile
Genocea Biosciences, Inc., incorporated on August 16, 2006, is harnessing the power of T cell immunity to develop vaccines and immunotherapies. The Company uses its discovery platform, AnTigen Lead Acquisition System (ATLAS), to design vaccines and immunotherapies that act, in part, through T cell (or cellular) immune responses.
The Company has one product candidate in Phase III clinical development, GEN-003, an immunotherapy for the treatment of genital herpes. It also has a pre-clinical immuno-oncology program focused on personalized cancer vaccines (GEN-009). The GEN-009 program leverages ATLAS to identify patient neoantigens, or newly formed antigens to each patient, that are associated with that individual’s tumor.
GEN-003
GEN-003 is an investigational T cell-directed immunotherapy designed to elicit both a T cell and B cell (antibody) immune response in patients with genital herpes. In multiple Phase 2 randomized, placebo-controlled clinical trials, GEN-003 has demonstrated results to date by showing statistically significant reductions in the clinical signs of genital herpes and viral shedding.
The immunotherapy designed for use of Genocea’s ATLAS platform, which profiled the comprehensive spectrum of actual T cell responses mounted by humans in response to HSV-2 to identify antigen targets that drive protective T cell responses to the virus. Reuters










