Sonoma Pharmaceuticals, Inc. (NASDAQ: SNOA)
Shares of Sonoma Pharmaceuticals, Inc. climbed more than 30% in premarket session on Thursday, following news that the company has received an approval from the US Food and Drug Administration for its antimicrobial skin gel.
The post-therapy gel is designed to treat itch and pain emanating from chemical peels and post-microdermabrasion therapy, as well as laser therapy procedures.
The gel is also ideal in the treatment of minor burns and skin irritations. Sonoma said that dermatology surgeons carried out over 9.5 million treatments in 2013, up almost 22 percent from the year before.
SNOA Earnings & Outlook
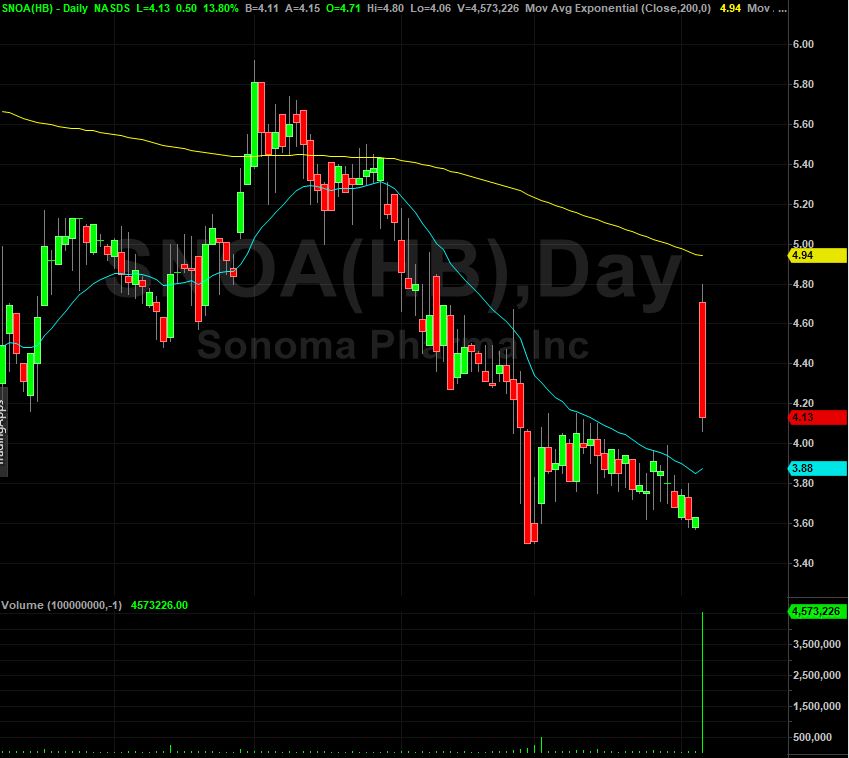
In the last twelve months, shares of the company have gone down about by 48%. Earnings are expected to go up by 89.65% over the next few years, indicating a very positive future ahead.
The earnings should lead to a higher share value, as a result of stronger cash flows. The company experienced a double-digit rise of more than 10% in its share price in the past several months on the Nasdaq Capital Market.
Sonoma’s CEO Comments
During the release of the company’s Q3 2018 results, the company’s Chief Executive Officer James Schutz said that product revenue had gone up by 46% compared to the prior-year period. “US product revenue grew by 27%, compared to the previous year’s quarter. Dermatology revenue increased by 78% compared to the previous year’s period,” Schutz said.
Sonoma Pharmaceuticals, Inc. Company Profile
Formerly known as Oculus Innovative Sciences, Sonoma Pharmaceuticals, Inc. is a drug manufacturer based in Petaluma, California. The company was founded in 1999, and develops and sells solutions used in the treatment of advanced tissue care and dermatological conditions.
Sonoma sells its products internationally and across the United States. Its products have improved the lives of over 5 million patients in various parts of the world by reducing pain, harmful inflammatory responses, infections, scarring and itch.
The company provides Microcyn products line, which is based on electrically charged molecules that are designed to deal with pathogens, such as bacteria, viruses, spores, antibiotic-resistant strains and fungi.
The Microcyn medical devices that the company makes are used for lubricating, cleaning, dressing, debridement, and moistening of chronic and acute sores in tissue care management. Other products made by the company include Ceramax Skin Barrier Cream, Celacyn, Mondoxyne, Alevicyn, and SebuDerm.
Sonoma Pharmaceuticals also operates a microbiology lab that offers laboratory and consulting services to companies in the biomedical sector. Its products are sold directly to end users, or available through distributors, hospitals, nurses, physicians, as well as other healthcare professionals.
company carries out its manufacturing operations in the US, Latin America and Europe. Its sales and marketing headquarters in Europe are located in Roermond, Netherlands. –Reuters










